News
The Employment Development Department, or EDD, said Lake County’s July unemployment rate was 7.4%, down from 7.5% in June and 11% in July 2020.
Leading the county’s job categories was total farm with a 6% growth over the month and 29.5% over the year. Leisure and hospitality was up by 2.5% over June and 17.5% over July 2020.
Nationwide, the jobless rate was down. The Bureau of Labor Statistics said July’s national unemployment rate was 5.4%, down from 5.9% in June and 10.2% in July 2020.
California’s unemployment rate was 7.6% in July, the same as June but down from 13.2% from July 2020.
California payroll jobs totaled 16,529,000 in July 2021, up 114,400 from June 2021 and up from 15,644,600 from July 2020.
Of the 2,714,800 jobs lost in March and April 2020 due to the COVID-19 pandemic, California has now regained 1,582,900 jobs, or 58.3%.
The state said the number of Californians employed in July was 17,493,600, an increase of 56,000 persons from June’s total of 17,437,600 and up 1,225,600 from the employment total in July of last year.
The number of unemployed Californians was 1,433,800 in July, a decrease of 5,800 over the month and down 1,029,700 in comparison to July of last year, the EDD reported.
Lake County’s jobless rate earned it a statewide rank of No. 36.
Neighboring county jobless rates and ranks for July are: Colusa, 10.6%, No. 55; Glenn, 7%, No. 31; Mendocino, 6.3%, No. 19; Napa, 5.8%, No. 11; Sonoma, 5.6%, No. 7; and Yolo, 5.9%, No. 15.
Marin continued to have the lowest unemployment of the state’s 58 counties, 4.5%, while Imperial County had the highest rate, 18.9%.
In related data, the EDD said there were 834,513 people certifying for Unemployment Insurance benefits during the July 2021 sample week. That compares to 592,867 people in June and 3,144,098 people in July 2020.
At the same time, 64,642 initial claims were processed in the July 2021 sample week, which was a month-over increase of 519 claims from June, but a year-over decrease of 179,864 claims from July 2020, the EDD reported.
Email Elizabeth Larson at
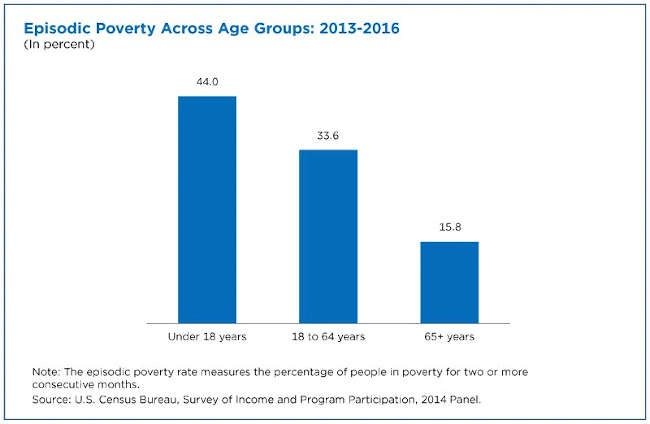
This rate was nearly triple the episodic poverty rate for individuals ages 65 and over.
The new report, Dynamics of Economic Well-being: Poverty, 2013-2016, includes longitudinal statistics like episodic and chronic poverty and the median length of poverty spells.
It shows the episodic poverty rate was higher (44%) for children (under age 18).
Seniors (ages 65 and over) had the lowest episodic poverty rate among all age groups with a rate of 15.8%. Working-age adults (ages 18-64) had a higher rate of 33.6% but still lower than the rate for children.
Chronic poverty
Across age groups, shorter poverty spells were much more common than multi-year poverty spells between 2013 and 2016.
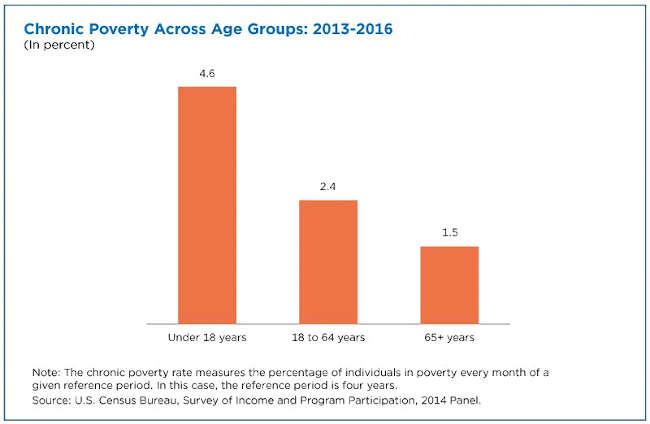
Chronic poverty describes individuals in poverty for all months of the 2013-2016 period studied. As a result, the chronically poor are also captured within the episodically poor, as they have more than two consecutive months in poverty.
As with episodic poverty, children also had the highest chronic poverty rate (4.6%) among all three age groups from 2013 to 2016. The chronic poverty rate for seniors was 1.5%, the lowest among all age groups, and 2.4% for working-age adults.
Lifting out of poverty
The length of a poverty spell represents the number of consecutive months an individual was in poverty. A group’s median poverty spell length can provide information on its ability to exit poverty.
Seniors and children typically remained in poverty longer than working-age adults, based on the median length of poverty spells by age group.
While seniors notably had the lowest episodic poverty rate among all age groups, those in poverty appeared to have difficulty exiting poverty.
Seniors’ median poverty spell length (12.4 months) was longer than it was for working-age adults (10.1 months) and was not statistically different than for children (11.8 months) from 2013 to 2016.
Abinash Mohanty is a statistician in the Census Bureau’s Social, Economic and Housing Statistics Division.
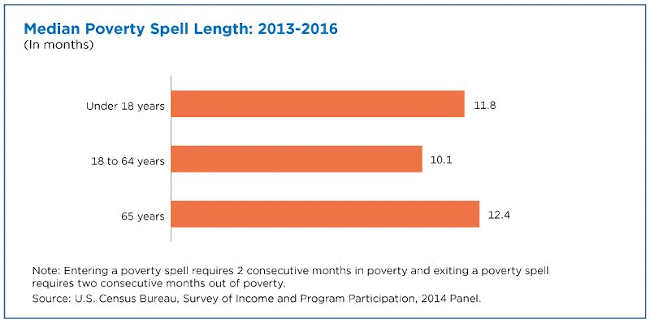
The board will meet beginning at 9 a.m. Tuesday, Aug. 31, in the board chambers on the first floor of the Lake County Courthouse, 255 N. Forbes St., Lakeport.
The meeting can be watched live on Channel 8, online at https://countyoflake.legistar.com/Calendar.aspx and on the county’s Facebook page. Accompanying board documents, the agenda and archived board meeting videos also are available at that link.
To participate in real-time, join the Zoom meeting by clicking this link.
The meeting ID is 935 3617 1596, pass code 598091. The meeting also can be accessed via one tap mobile at +16699006833,,93536171596#,,,,*598091# US.
All interested members of the public that do not have internet access or a Mediacom cable subscription are encouraged to call 669-900-6833, and enter the Zoom meeting ID and pass code information above.
To submit a written comment on any agenda item visit https://countyoflake.legistar.com/Calendar.aspx and click on the eComment feature linked to the meeting date. If a comment is submitted after the meeting begins, it may not be read during the meeting but will become a part of the record.
In an item timed for 11 a.m., the board will hold a public hearing to consider extending an interim urgency ordinance establishing a moratorium on the issuance of early activation permits on all land use projects in the unincorporated county.
The board unanimously approved the ordinance for a 45-day period in July, as Lake County News has reported.
The extension would be for 10 months and 15 days.
In other business on Tuesday, at 9:06 a.m. the board will get a Lake County schools COVID-19 update from Superintendent of Schools Brock Falkenberg.
At 10 a.m., the board will hold its first public hearing to introduce the redistricting process for supervisorial districts.
At 10:30 a.m., the board will consider its response to the 2020-21 Grand Jury Final Report.
Other timed items include the agreement for an operational analysis of indigent defense services set for 11:30 a.m. and presentation of the Roadmap Task Force Pilot Project at noon.
The full agenda follows.
CONSENT AGENDA
5.1: Appoint temporary representatives to meet for unrepresented Confidential A and B employees regarding salary and benefits.
5.2: Adopt resolution amending Resolution No. 2021-68 Establishing Position Allocations for Recommended Fiscal Year 2021-2022 Budget, Budget Unit No. 2702, Planning.
5.3: Approve Amendment No. 1 to agreement for recruitment services for the Lake County Public Health officer.
5.4: Adopt resolution approving Agreement No.21-0341-000-SA with California Department of Food and Agriculture for Compliance with the Nursery Inspection Program for Period July 1, 2021, through June 30, 2022, in the amount of $990.
5.5: Sitting as Lake County Air Quality Management District Board of Directors, authorize the air pollution control officer to sign and submit the grant agreement between the district and ARB for AB617 Implementation Funds for the Community Air Protection Program, and sign all other program documents.
5.6: Approve Board of Supervisors meeting minutes for July 20, 2021.
5.7: Adopt resolution (a) authorizing entering into a funding agreement with the State Water Resources Control Board and (b) authorizing and designating Lake County Special Districts administrator to sign, on the behalf of CSA-21, North Lakeport Water, all grant related documents for the Drought Resiliency Project.
TIMED ITEMS
6.2, 9:06 a.m.: Lake County schools COVID-19 update.
6.3, 10 a.m.: Redistricting public hearing No. 1 — Introduction to the redistricting process.
6.4, 10:30 a.m.: Consideration of response to the 2020-21 Grand Jury Final Report.
6.5, 11 a.m.: Public hearing, consideration of an ordinance extending interim Urgency Ordinance No. 3107 10 months and 15 days which establishes a moratorium on the issuance of early activation permits for land use projects within the unincorporated area of Lake County and authorize chair to sign .
6.6, 11:30 a.m.: Consideration of agreement for operational analysis of indigent defense services.
6.7, noon: Presentation of Roadmap Task Force Pilot Project.
UNTIMED ITEMS
7.2: (a) Consideration of a Presentation on PG&E’s Better Together Resilient Communities Grant Program; (b) consideration of a resolution of support for Trane Technologies to submit an application on behalf of the county of Lake to PG&E’s “Better Together Resilient Communities” Grant Program, in pursuit of funds for additional engineering and due diligence in the evaluation of potential advanced energy projects.
CLOSED SESSION
8.1: Public employee evaluation: Public Works Director Scott De Leon.
8.2: Public employee evaluation: Behavioral Health Director Todd Metcalf.
8.3: Public employee appointment pursuant to Gov. Code Section 54957(b)(1): (a) Appointment of Community Development director.
8.4: Conference with temporary representatives designated to meet with county department heads regarding salary and benefits of unrepresented management employees.
Email Elizabeth Larson at
Drug overdose is now the No. 1 cause of unintentional injury-related death in the United States.
In 2019, more than 70,000 people died from drug overdoses, over 6,000 of those here in California.
The leading driver of these deaths (greater than 70%) is opioid drugs, including fentanyl, heroin and prescription opioids.
Lake County's death rate due to drug use is more than four times higher than the state of California’s.
To help raise community awareness about drug overdose, Lake County Behavioral Health Services is shining a spotlight on it through International Overdose Awareness Day.
This is a global event which takes place annually on Aug. 31, to remember those gone too soon from drug overdose deaths.
A tribute event will take place on Tuesday from 10 a.m. to 5 p.m., with speakers at 12:30 p.m. in Lakeport’s Library Park.
Individuals who consume prescription opioids, even when following the prescribed dose, may eventually develop a tolerance to the drug.
When pain has decreased or gone away, some folks find it easy to stop taking the drugs. Others find it difficult to quit. Those who find it harder to quit may continue to take opioids longer than necessary; worse, they may develop an opioid use disorder.
People prescribed opioid pain relievers are also at risk of developing an opioid use disorder. Many are unaware their opioid use has become problematic.
The United States Department of Health and Human Services’ Substance Abuse and Mental Health Services Administration reports over half of the people who have misused prescription drugs claimed they got the drugs from relatives or friends. Most people do not know sharing opioids is a felony.
People who take pain relieving drugs in doses too large or for an extended period of time are more at risk of developing an opioid use disorder, and face greater risk of death due to opioid overdose.
Nearly 10 million people over the age of 12 misused prescription drugs in 2019, with 2 million of those having opioid use disorder, per the National Safety Council.
The good news is opioid use disorders are treatable, and recovery can be achieved with effective treatment. Many people in recovery go on to live healthy, successful lives.
For more information, please contact Lake County Behavioral Health Services at 707-274-9101 or 707-994-7090.
If you or someone you know is in urgent need of help, please call the county’s 24-hour crisis line at 800-900-2075.

Thirty percent of unvaccinated American adults say they’re waiting for the COVID-19 vaccines currently authorized for emergency use to be officially approved by the U.S. Food and Drug Administration. The FDA has since granted that approval for those age 16 and older for the Pfizer-BioNTech vaccine on Aug. 23, 2021. What had to happen for the FDA to advance from emergency use authorization, or EUA, to full approval?
I’m a pharmacist who trains other pharmacists, health care providers and students on why, when and how to administer vaccines. Emergency use authorization, while streamlining the regulatory process so the vaccine is more quickly available to the public, still follows a rigorous process the FDA requires to ensure vaccine safety and effectiveness. The difference is that more time has passed and more data is available for review when a company applies for full approval.
EUA and full approval share similar first steps
For both emergency authorization and full approval, for COVID-19 vaccines, the FDA first requires initial safety studies on a small number of people. Here, researchers document potential adverse events, or side effects, that the vaccine may cause. Researchers also determine the safest and most effective dose of the vaccine.
Once the vaccine is determined to be safe and an optimal dose identified, researchers will create larger studies to ascertain how well it works in a controlled setting where some people are given the vaccine while others are given a placebo.
It is important to note that the number of people who participated in the initial COVID-19 safety studies was similar to that in the safety studies of other commonly used vaccines, including vaccines for tetanus, diphtheria, whooping cough and meningitis. Over 43,000 adults participated in the early phases of the Pfizer-BioNTech clinical trial, over 30,400 in Moderna’s and over 44,000 in Johnson and Johnson’s. Half the participants in each study were given a vaccine, while the other half were given a placebo.
Where EUA and full approval differ
From this point on, emergency use authorization and full FDA approval for COVID-19 vaccines follow different clinical study requirements.
For the emergency use authorization, the FDA requires at least half of the participants of the original studies to be followed for at least two months post-vaccination. This is because the vast majority of vaccine-related side effects occur right after vaccination.
Full FDA approval, on the other hand, requires participants in the original studies to be followed for at least six months. Reviewers look at data from the same study participants but collected over a longer period of time. All adverse events are examined. The manufacturer must also provide more detailed manufacturing plans and processes, as well as a higher level of oversight and inspections. All of this adds significantly more time to the review process.

Both Pfizer and Moderna began their rolling submission for approval in the FDA’s “Fast Track” process, designed to speed up review. This allows the companies to submit portions of their approval application to the FDA for review as they’re completed.
Full FDA approval will initially apply to only the same age groups that were tested in the original emergency use authorization. This means that the vaccine has been approved first for people ages 16 and up for Pfizer, and will likely be approved for those 18 and up for Moderna. Rolling submission will allow approval of the vaccine for younger groups as more data becomes available.
Different timelines, same rigorous requirements
Full FDA approval is a milestone that may help build confidence among the vaccine hesitant about the safety of the vaccines. But the true test of the vaccine came when it first gained emergency use authorization. Then, researchers identified the majority of its potential side effects and proved its ability to protect against severe disease.
Article updated on Aug. 23, 2021 to reflect FDA approval of Pfizer BioNTech vaccine for those ages 16 and older.
[Like what you’ve read? Want more? Sign up for The Conversation’s daily newsletter.]![]()
Jennifer Girotto, Associate Clinical Professor of Pharmacy Practice, University of Connecticut
This article is republished from The Conversation under a Creative Commons license. Read the original article.
The following cats at the shelter have been cleared for adoption.
Call Lake County Animal Care and Control at 707-263-0278 or visit the shelter online at http://www.co.lake.ca.us/Government/Directory/Animal_Care_And_Control.htm for information on visiting or adopting.

Female domestic short hair kitten
This female domestic short hair kitten has a black coat.
She is in cat room kennel No. 53, ID No. LCAC-A-1503.

Domestic medium hair cat
This 3-year-old female domestic medium hair cat has a brown tabby coat.
She is in cat room kennel No. 58, ID No. LCAC-A-1029.

‘Marmalade’
“Marmalade” is a 5-year-old female domestic short hair cat with a calico and white coat.
She is in cat room kennel No. 68, ID No. LCAC-A-1444.

Female domestic short hair kitten
This female domestic short hair kitten has a black coat.
She is in cat room kennel No. 101, ID No. LCAC-A-1504.

Domestic short hair kitten
This male domestic short hair kitten has all-black coat.
He is in cat room kennel No. 101, ID No. LCAC-A-1502.

Male domestic short hair
This male domestic short hair has a gray and white coat.
He is 1-year-old and weighs nearly 6 pounds.
He is in cat room kennel No. 120, ID No. LCAC-A-874.

Female domestic short hair
This 2-year-old female domestic short hair cat has a white coat and blue eyes.
She is in cat room kennel No. 123, ID No. LCAC-A-1152.

Domestic short hair kitten
A male kitten from this litter remains available for adoption.
He is in cat room kennel No. 125B, ID No. LCAC-A-1139.

Female domestic short hair
This 1-year-old female domestic short hair cat has a black coat.
She is in cat room kennel No. 135, ID No. LCAC-A-1133.

‘Goldie’
“Goldie” is a male domestic short hair kitten with a yellow tabby and white coat.
He is in cat room kennel No. 142, ID No. LCAC-A-1442.

‘Ophir’
“Ophir” is a male domestic short hair kitten with a red and white coat.
He is in cat room kennel No. 142, ID No. LCAC-A-1443.
Email Elizabeth Larson at
 How to resolve AdBlock issue?
How to resolve AdBlock issue? 